My goal with this forum is to explain medicine and medical literature to those outside the medical field. The jargon alone can make the medical field difficult to understand and that is even more true for medical literature and study data. The vaccine data is a great example of this. If you have never worked with the software used to collect data for clinical studies, the printouts can be difficult to follow. I hope to help with this while providing my interpretation as I go.
Source: All files downloaded from phmpt.org. I had hoped to find the files on the FDA’s site but was unable to at the time of this writing. PHMPT is the group that sued the FDA for the release of these files. While they may be biased regarding the goal of the lawsuit, this seems to be the best available source to download all of the released files.
My Roadmap
The first step I plan to take is to catalog what each file contains. When posted, this will appear as the file name followed by one or two sentences briefly describing what is contained in the file. I will also do more detailed descriptions of files of interest. This may take the form of a bulleted description with page numbers (such as I provide below) or a narrative discussion when warranted. As always, I will take requests. If there is a specific file you would like me to go through early, let me know.
The First Five Files
When I downloaded the files, I placed them into folders based on the month they were released. These then got automatically organized in the folder by numerical/alphabetical order. I apologize if this order doesn’t match the order of the files online.
These first five files are participant data reports that contain all of the collected information on these 5 study participants. Each file contains >70 pages. If the releases will contain the forms for every participant, that could partially explain the enormous volume of pages to be released.
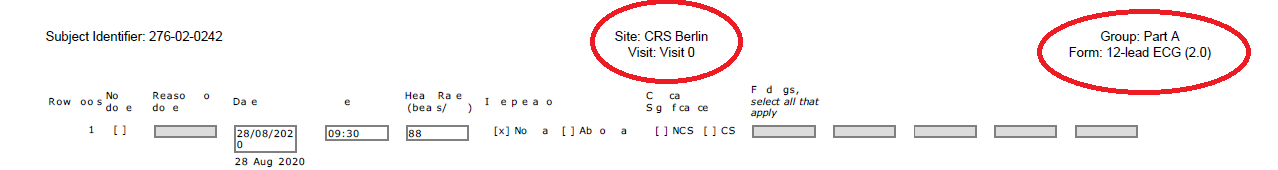
Each file has a header that looks like this. The top left is the subject identifier which shows the participant’s study ID. Each of the files I have looked at contains information for a single study participant, so this number is the same throughout the file. The middle part of the header (circled) shows the study site and the study visit number. Each participant had multiple study site visits which are why there are a lot of similar appearing forms. The far-right (circled) tells you the name of the form, which typically will explain what information you are looking at on that page.
Without further ado, here is what is in the first 5 files I reviewed:
125742_S1_M5_5351_bnt162-01_10010 – 75 pages - Study data for a single participant that had an adverse reaction and did not complete the entire study protocol.
Page 60 - shows local symptoms experienced and severity
Page 62 - shows systemic symptoms experienced and severity
Page 65 - shows new medications taken by the participant during the study period (ibuprofen, acetaminophen (paracetamol is the European equivalent), and local anesthetic injection (similar to lidocaine))
Page 72 - shows a negative COVID test
125742_S1_M5_5351_bnt162-01_10075 – 94 pages - Study data for a single participant that completed the study protocol but had an adverse reaction
Page 27 - shows a negative COVID test
Page 84 - shows all reported symptoms
Page 86 - shows local symptoms experienced and severity
Page 87 - shows systemic symptoms experienced and severity
Page 90 - shows new medications taken by the participant during the study period (acetaminophen (paracetamol is the European equivalent), topical dontisolon (similar to hydrocortisone cream), Canesten (antifungal cream), and local anesthetic injection (similar to lidocaine))
125742_S1_M5_5351_bnt162-01_20116 – 81 pages - Study data for a single participant that did not complete the entire study protocol due to missing a visit because of illness and then was unable to receive a second dose due to a prolonged period from the first dose.
Page 26, 36, 45, 54, and 62 - show local injection site symptoms
Page 27 - shows a negative COVID test
Page 69 - shows all reported symptoms
Page 70 - shows a description of reasons for not completing the trial
Page 71 - shows local symptoms experienced and severity
Page 72 - shows systemic symptoms experienced and severity
Page 77 - shows new medications taken by the participant during the study period (Arnica (herbal supplement), Ruta (herbal supplement), Bromelain (dietary supplement), Tyrosur (topical antibiotic), Gelomyrtol (mucolytic like Mucinex), and Vagisan (cream used for vaginal dryness))
Page 78 - shows protocol deviations
125742_S1_M5_5351_bnt162-01_20215 – 97 pages – Study data for a single participant that experienced a serious adverse event that is almost certainly unrelated to the study but would still be reported in the adverse event report for the study.
Page 26, 47, 57, 68, and 76 - show local symptoms experienced and severity (none in this case)
Page 27 - shows a negative COVID test
Page 30, 40, 50, 60, 71, and 79 - show systemic symptoms experienced and severity (none in this case)
Page 84 - shows serious adverse event (patient broke their ankle and required hospitalization)
Page 85 - shows further information about the event (patient slipped getting on a train and broke their ankle)
Page 86 and 87 - show self-reported local symptoms from the participant’s symptom diary
Page 88 - shows self-reported systemic symptoms from the participant’s symptom diary
Page 91 - shows new medications taken by the participant during the study period (pantozol (antacid, equivalent to Protonix), Valoron (opioid pain reliever), Ibuprofen, clexane (anticoagulant, used to prevent blood clots are the broken ankle), Tilidin (opioid pain reliever, generic of Valoron)
125742_S1_M5_5351_bnt162-01_20242 – 100 pages - Study data for a single participant that did not receive their second dose because they passed out during the visit before administration of the dose
Page 26, 37, 47, 57, 68, 76, and 84 - show local symptoms experienced and severity
Page 27 - shows a negative COVID test
Page 30, 40, 50, 60, 71, 79, and 87 - show systemic symptoms experienced and severity
Page 90 - shows adverse events reported. The participant had chest wall pain and passed out
Page 91 - shows comments, including that participant had headache and chills after injection
Page 92 - show self-reported local symptoms from the participant’s symptom diary
Page 93 - shows self-reported systemic symptoms from the participant’s symptom diary
Page 96 - shows new medications taken by the participant during the study period (acetaminophen (paracetamol is the European equivalent) and Novalgin (non-steroidal anti-inflammatory that is banned in the US due to adverse events)
Conclusion
These files are just the tip of the iceberg, but they do raise some interesting ideas. The first is that both local and systemic symptoms are common (known but useful to see documented). The second is that just because an adverse event is reported in a study does not mean that it is related to the drug/vaccine. There is a big difference between slipping on a step and breaking your ankle (the case in the 4th file) and having weakness leading to a fall and broken ankle. The first is not related to the study while the second very well could be. These both get reported as serious adverse events and it is left to adjudicators to determine if it is related to the study or not. The patient that passed out is even more complicated. They had not received the injection yet during that visit and there is very little other information available in this file. These cases highlight why simply looking at a list of adverse events in a study tells you very little about the safety of a drug/vaccine.
I think this journey through the Pfizer documents will be enlightening. If nothing else, it will provide a clear view of how clinical trials are done and the way that data is reported.